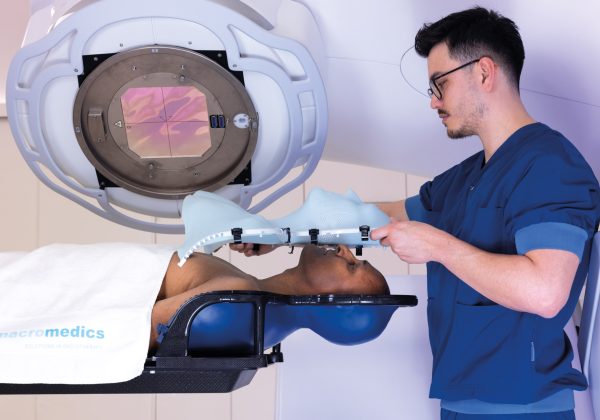
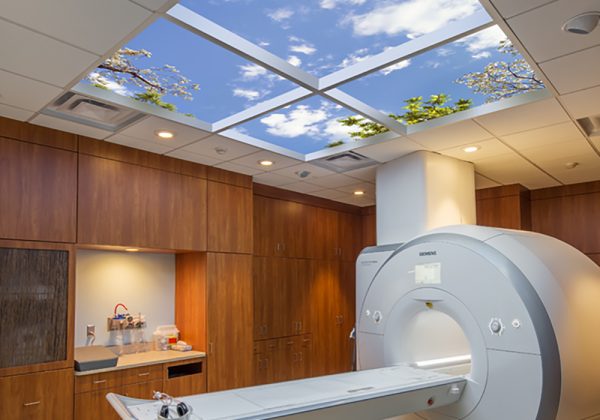
- Clinical
- Physics
- QA
- Dosimetry
- Proton QA & Dosimetry
- Software
- ART-Plan by TheraPanacea
- COMPASS Patient Dose QA
- ImSimQA™
- myQA® Software Physics QA Platform
- myQA® iON for conventional radiotherapy
- myQA® iON for Proton Therapy
- ProSoma – Virtual Simulation
- ProSoma Core – MC plan, check, and logfile analysis
- RadCalc QA Software
- RIT Family of Products
- SagiPlan® HDR & Focal Therapy Planning
- MRI Physics Solutions
- SGRT
- Medical Imaging
- Exterior and Interior Design
- Manufactured by
- Support & Finance
- News
- About
- Contact


Adaptive. Innovative. Reliable.
Oncology Systems are suppliers of outstanding products, services, support and value to the radiotherapy community.
How may we help?
Featured products and services
Oncology Systems (OSL) specialises in radiotherapy and oncology technology for the treatment of cancer.
We distribute leading-edge medical technology from state-of-the-art medical imaging, treatment planning and AI-based auto contouring software, external beam and brachytherapy treatment systems, medical physics devices, to radiotherapy patient positioning systems and accessories.
OSL can also assist with shielding and SkyCeiling lighting solutions for the exterior and interior of healthcare buildings.
OSL supplies products, equipment, and software to the UK and Ireland radiotherapy centres.
News & Events
- UK & Ireland Prostate Brachytherapy Conference 2024 28th February 2024
- IAPM ASM 2024 28th February 2024
- ESTRO 2024 12th January 2024
Support

Expert. Responsive. Tailored.
OSL strives to provide expert and professional support to our customers in a range of ways, from installation and ongoing maintenance agreements through to training, commissioning services and user meetings and symposia.
 Latest tweets
Latest tweets
📅 Save the dates! We look forward to seeing you for the relaunch of the Annual Radiotherapy Conference later this year. Register your interest here to receive information as soon as it's available: https://bit.ly/SORARC24
Please do get in touch if you’d like to secure your spot to appear on the podcast! We are revamping our podbox this year, so it’ll be the place to be!! Make sure to add it to your conference agenda @UKIOCongress @EmmaHydeTeach @rijan44 @RCRadiologists @SCoRMembers @SoRStudentReps
👀 Join the team!
We have a job opportunity for an Account Specialist in Radiotherapy Sales. Please read the details on our website.
***NEW MEMBERS ANNOUNCEMENT *** @_AXREM are pleased to advise that this week we have welcomed @OSL1 to membership and they will input into the great work being done by our Radiotherapy Special Focus Group. Find out more about OSL here: https://www.axrem.org.uk/member/oncology-systems-limited/
📢Registration opens 1 April for our two-day Magnetic Resonance Guided #Radiotherapy (MRIgRT) course
📅 Mon 23-Tues 24 Sept
📍ICR and @royalmarsdenNHS Sutton
Find out more about this #Training and register👇
https://www.icr.ac.uk/studying-and-training/opportunities-for-clinicians/radiotherapy-and-imaging-training-courses/magnetic-resonance-image-guided-radiotherapy-(mrigrt)
NELCO Worldwide has completed building an IMPERVIUS™ Megashield™ door-less maze entry radiotherapy treatment room at Mid and South Essex NHS Foundation Trust Southend University Hospital.
The custom build included a maze entry design to eliminate the need for a shielding door.
We are thrilled to have joined @AXREM and look forward to collaborating with the team. #radiotherapy #oncology
OSL and LAP team up to bring patient-friendly SGRT to UK and Ireland.
Full story now available online:
@OSL1
#RADMagazine #medicalimaging #news #healthcare #medical #radiotherapy
Having a fantastic day of learning about particle physics and accelerators, including aspects of medical physics and Radiotherapy! Had brilliant talks and activities from experts.....here is Rof Julia Handley teaching about proton therapy!
We are so thrilled to have an amazing set of speakers appearing in our Rad Chat #radiotherapy theatre at @oncology_care. Take a look at the overall programme and regsiter for this FREE conference for healthcare professionals working in oncology. #conference #oncology
We are excited about the upcoming exhibition and cannot wait to meet and catch up with everyone!
📅 Save the dates! We look forward to seeing you for the relaunch of the Annual Radiotherapy Conference later this year. Register your interest here to receive information as soon as it's available: https://bit.ly/SORARC24
Please do get in touch if you’d like to secure your spot to appear on the podcast! We are revamping our podbox this year, so it’ll be the place to be!! Make sure to add it to your conference agenda @UKIOCongress @EmmaHydeTeach @rijan44 @RCRadiologists @SCoRMembers @SoRStudentReps
👀 Join the team!
We have a job opportunity for an Account Specialist in Radiotherapy Sales. Please read the details on our website.
***NEW MEMBERS ANNOUNCEMENT *** @_AXREM are pleased to advise that this week we have welcomed @OSL1 to membership and they will input into the great work being done by our Radiotherapy Special Focus Group. Find out more about OSL here: https://www.axrem.org.uk/member/oncology-systems-limited/
📢Registration opens 1 April for our two-day Magnetic Resonance Guided #Radiotherapy (MRIgRT) course
📅 Mon 23-Tues 24 Sept
📍ICR and @royalmarsdenNHS Sutton
Find out more about this #Training and register👇
https://www.icr.ac.uk/studying-and-training/opportunities-for-clinicians/radiotherapy-and-imaging-training-courses/magnetic-resonance-image-guided-radiotherapy-(mrigrt)
NELCO Worldwide has completed building an IMPERVIUS™ Megashield™ door-less maze entry radiotherapy treatment room at Mid and South Essex NHS Foundation Trust Southend University Hospital.
The custom build included a maze entry design to eliminate the need for a shielding door.
We are thrilled to have joined @AXREM and look forward to collaborating with the team. #radiotherapy #oncology
OSL and LAP team up to bring patient-friendly SGRT to UK and Ireland.
Full story now available online:
@OSL1
#RADMagazine #medicalimaging #news #healthcare #medical #radiotherapy
Having a fantastic day of learning about particle physics and accelerators, including aspects of medical physics and Radiotherapy! Had brilliant talks and activities from experts.....here is Rof Julia Handley teaching about proton therapy!
We are so thrilled to have an amazing set of speakers appearing in our Rad Chat #radiotherapy theatre at @oncology_care. Take a look at the overall programme and regsiter for this FREE conference for healthcare professionals working in oncology. #conference #oncology
We are excited about the upcoming exhibition and cannot wait to meet and catch up with everyone!